Function
Proteins carry out numerous roles, and are involved in virtually all life processes in biological organisms
Introduction
Proteins are perhaps the most interesting molecules in life sciences.
Why is this the case? First, they are the most abundant macromolecules in cells, making up as much as ~50% of the cell’s total dry mass. Moreover, the number of different functional proteins in cells and tissues is much higher than the number of other macromolecules.
Although RNA molecules have been discovered in recent years to be much more diverse than previously believed and to carry out certain cellular functions such as regulation and catalysis, they have yet to match the diversity of proteins.
Protein diversity is particularly pronounced in eukaryotic cells, due to gene splicing and post-translational processing.
For example, the human body is estimated to contain only 20,500 genes but ~100,000 different proteins, and biochemical methods of protein detection suggest that each cell may express up to 15,000 distinct proteins.
The total number of protein types in nature has yet to be determined, although estimates do exist.
For example, in specific organisms whose genomes have been sequenced, it is possible to determine the total number of different proteins according to the number of open reading frames in the organisms’ DNA.
The number of proteins produced thus far by this method is on the order of millions.
Another estimate, carried out according to the (estimated) number of species on Earth, suggests a much greater number of proteins: 1010 – 1012.
In any case, these estimates indicate that large numbers of proteins exist in nature.
This conclusion is highly significant, as it suggests that proteins are functionally diverse.
Catalysis of metabolic processes
Living organisms maintain a wide range of metabolic processes that allow them to grow or divide, extract energy from foodstuff, build complex materials, decompose waste products, detoxify harmful substances, etc.
__ These metabolic processes, which are responsible for sustaining life in all organisms, involve thousands of chemical reactions that cells and tissues execute both simultaneously and consecutively__.
Many of these reactions occur readily, as their products are more stable (i.e., have less free energy*) than their reactants. However, the molecular needs of the organism dictate that these reactions must be completed within a timescale of 10−5 to 102 seconds.
In stark contrast, many chemical reactions have much longer half-lives, which may span from minutes to millions of years.
During their long evolution processes, living organisms have developed means of speeding up the chemical reactions occurring within them.
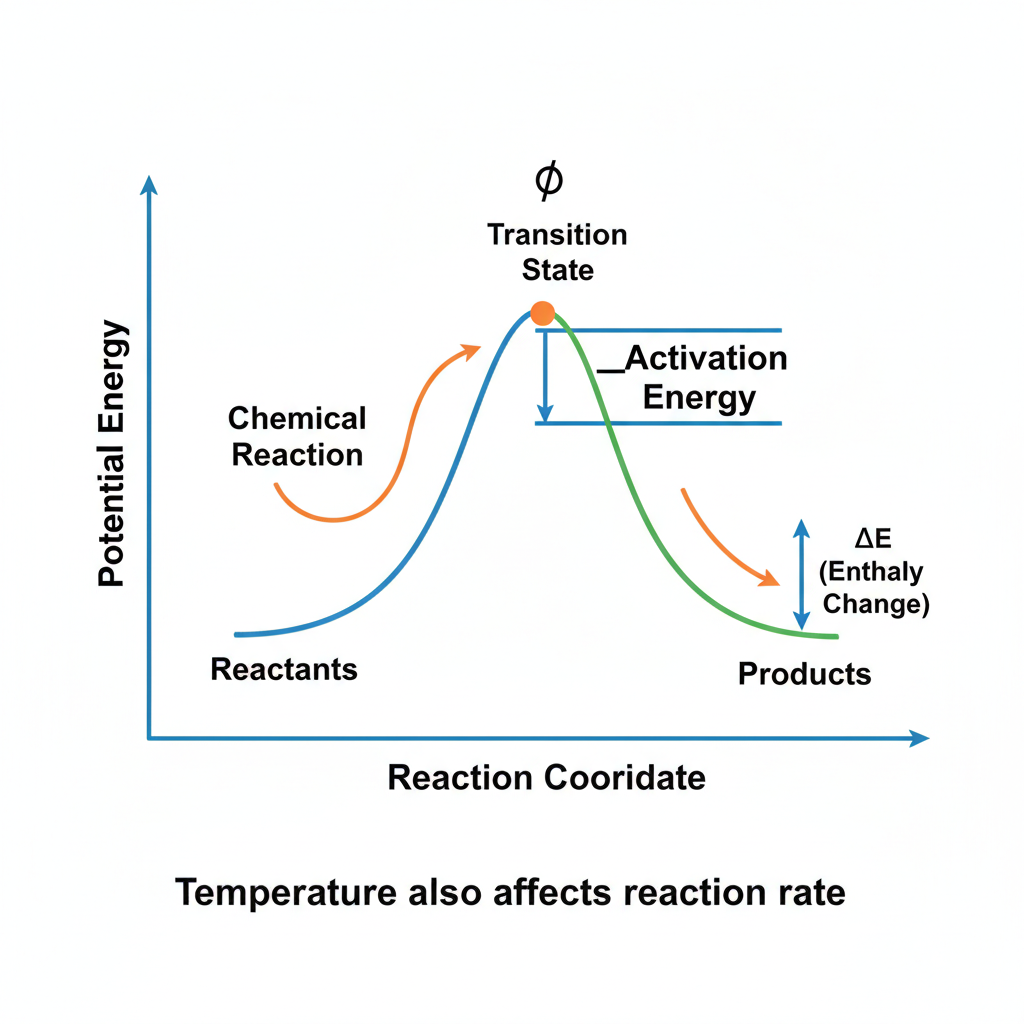
According to the currently accepted model, a chemical reaction transforms reactants into products via a short-lived and high-energy transition state.
The energy required for a reactant to be converted into its transition state is called the activation energy , and the rate of the reaction depends directly on the magnitude of this energy, as well as on the temperature.
This relationship is captured by the well-known Arrhenius equation:
= 8.314462618 # J/(mol*K)
"""Compute rate constant k using the Arrhenius equation."""
return *
# Example:
= 1.0e13 # 1/s (example units)
= 75_000.0 # J/mol
= 298.15 # K
=
(where 𝑘 is the reaction rate, 𝑅 is the universal gas constant (1.989 cal/(molK)), and 𝑇 is the absolute temperature (in K)).
In other words, the reactant(s) must gain energy that is equal to or higher than the activation energy, to be able to reach the transition state and turn into product(s).
One way to enable a reactant to gain sufficient activation energy is to increase the temperature to a very high value. This approach is infeasible in living organisms, however, as they exist and function within a very limited temperature range. Thus, the only way for an organism to accelerate its metabolic reactions is to lower the activation energy required, i.e., to stabilize the transition states of its reactions.
This can be done by catalysts, i.e., chemical species that accelerate reactions without changing themselves.
Different elements, such as transition metals, may serve as catalysts. Many of these chemical species are available in the various environments on our planet, and could therefore be easily harvested by living organisms. Surprisingly, however, biological organisms did not adopt these chemical catalysts.
Enzymes
Organisms have developed a wide range of proteins that can accelerate (catalyze) chemical reactions. These catalytic ‘nanomachines’ are termed enzymes.
Enzymes are highly selective catalysts: they typically speed up a reaction only for particular substrate(s), even when many other molecules are present
Why were enzymes selected over simple catalysts during evolution? There are several possible reasons, but the most likely is the high specificity of enzymes towards their intended reactants (a.k.a. substrates).
In contrast to simple catalysts such as metals, which can accelerate many different chemical reactions, each enzyme accelerates only a specific type of reaction, involving a specific substrate or substrates.
The specificity results from the complex three-dimensional structure of the enzyme; this structure includes an active site that is specifically designed to bind only the enzyme’s intended substrate (via noncovalent interactions), as well as to execute only the intended reaction.
The amazing specificity of enzymes constitutes a huge advantage in the highly diverse chemical environment within living organisms; it allows them to control the rates of each of their metabolic reactions by controlling the enzymes executing them.
Why the enzyme acts only on its intended substrate
This selectivity comes from the enzyme’s three-dimensional structure, especially the active site, which provides:
-
Geometric (shape) complementarity. The intended substrate fits the active site like a key in a lock (or, more accurately, the enzyme can slightly adjust—induced fit—to snugly accommodate the right substrate).
-
Chemical complementarity. Correct substrates make the right noncovalent interactions (e.g., hydrogen bonds, ionic interactions, hydrophobic contacts) with active-site residues. “Wrong” molecules usually cannot make enough of these interactions in the correct arrangement.
-
Catalytic positioning. Even if another molecule binds weakly, the enzyme generally won’t catalyze a reaction unless the substrate is held in the precise orientation needed to stabilize the transition state and place reactive bonds where the catalytic residues can act.
Energy transfer
Cells and tissues carry out a diverse set of processes, which are needed for sustaining life in all organisms. These processes include molecular biosynthesis, transport of chemicals across biological membranes, and movement. Most of these processes are not spontaneous and require an input of energy in order to take place.
This means that cells must be able to harness environmentally available energy in order to stay alive. Indeed, a large portion of each cell’s metabolic activity is dedicated to extracting and processing energy from available sources.
Despite being metabolically complex, all living organisms use at least one of two available sources of energy: chemical (i.e., food), and electromagnetic (i.e., solar radiation).
In both cases, a complex, multi-component machinery within the cell is responsible for utilizing the available energy source. Some of the components of this machinery are enzymes performing catabolic reactions, in which foodstuff molecules are simultaneously degraded and oxidized to extract the chemical energy stored in them.
However, there are other protein components that function in the efficient transfer of the extracted energy between different cellular compartments, or along a certain distance within a single compartment. These proteins are not enzymes, as they transfer energy in its pure form (i.e., as electrons or electromagnetic radiation), without causing any chemical change. The energy is finally stored as ATP, which is the most accessible form of energy in biological systems, and is therefore often referred to as the ‘universal energy currency’.
However, the utilization of raw energy from the source to form ATP is done gradually (for regulation purposes) using different energy-converting processes.
Defensa
Les cèl·lules contenen diferents mecanismes per combatre els patògens invasors.
Alguns d’aquests mecanismes són simples i es basen en enzims que reconeixen i destrueixen les molècules estranyes (per exemple, l’ARN de doble cadena, que només és present en els virus).
En els animals, llargs processos evolutius han conduït al desenvolupament d’un sistema de defensa altament eficient contra els patògens. Aquest sistema s’anomena “sistema immunitari” perquè és capaç de recordar els patògens que ja ha trobat i reaccionar tan ràpidament que l’animal infectat ni tan sols s’adona que ha estat compromès.
El sistema immunitari consta de dues branques.
-
La primera, la branca humoral, es basa principalment en els anticossos, molècules formades per glòbuls blancs anomenats limfòcits B, que patrullen el cos a través del sistema circulatori i localitzen elements estranys interactuant-hi específicament.
-
La segona branca, anomenada “branca cel·lular”, inclou els limfòcits T (alguns dels quals s’han esmentat més amunt). Aquests són glòbuls blancs formats a la medul·la òssia i es poden trobar al sistema circulatori, al sistema limfàtic, als teixits i en alguns fluids corporals (per exemple, la llet).
L’eficiència del sistema immunitari es deu a l’especialització de cadascun dels seus components i a l’ajustada cooperació entre ells.
Molts dels components del sistema immunitari són proteïnes, incloent-hi el TCR, els anticossos i les molècules MHC. Curiosament, aquestes molècules funcionalment diferents pertanyen al mateix grup de proteïnes, anomenat “immunoglobulines”. Les proteïnes que pertanyen a aquest grup tenen la mateixa estructura principal.
A més del TCR i el MHC, el grup de les immunoglobulines també inclou altres molècules unides a la membrana dels limfòcits, que ajuden a regular la resposta immunitària.
La branca humoral del sistema immunitari inclou, a més dels anticossos, un conjunt de proteïnes conegudes com a “sistema del complement”. Igual que els anticossos, les proteïnes del sistema del complement es troben a la sang i s’uneixen als patògens. Tanmateix, la resta del procés és diferent del cas dels anticossos: després d’unir-se al patogen, les proteïnes del complement s’uneixen entre elles i formen un complex letal. Aquest complex actua com un trepant molecular que perfora la membrana del patogen o bé en provoca la mort per altres mitjans indirectes.
Finalment, les proteïnes també intervenen en la comunicació a llarga distància entre les cèl·lules immunitàries i entre aquestes i altres cèl·lules del cos. Aquestes proteïnes, anomenades “citoquines”, són missatgers químics secretats per limfòcits activats o per altres glòbuls blancs. No només transmeten missatges entre limfòcits, sinó que també potencien les capacitats antivíriques d’altres cèl·lules (per exemple, en el cas de la citoquina anomenada “interferó”).
Importància mèdica.
A la taula següent s’enumeren algunes de les proteïnes més comunes dels eśsers vius, classificades pels tipus de funcions que exerceixen:
| Funció | Exemples | Descripció |
|---|---|---|
| Enzim digestiu | Amilasa, lipasa, pepsina | Degrada els nutrients als aliments en trossos més petits que poden ser absorbits fàcilment |
| Transport | Hemoglobina | Transporta substàncies pel cos a la sang o limfa |
| Estructura | Actina, tubulina, queratina, col·lagen | Forma diferents estructures, com el citoesquelet |
| Senyalització hormonal | Insulina | Coordina l’activitat de diferents sistemes del cos |
| Defensa | Anticosos | Protegeix el cos de patògens externs |
| Contracció | Miosina | Duu a terme la contracció muscular |
| Emmagatzematge | Proteïnes d’enmagatzematge en verdures, clara de l’ou (albúmina) | Proporciona aliment per al desenvolupament primerenc de l’embrió o la plàntula |
Com podeu veure, totes fan funcions fonamentals per preservar la salut dels éssers vius: les cel·lules, teixits, organismes…
Per això, moltes proteïnes són dianes farmacològiques importants per a molts medicaments.
Per exemple, els inhibidors de l’enzim convertidor d’angiotensina (ECA) s’utilitzen per tractar la hipertensió arterial, ja que bloquegen l’activitat de l’ECA, una proteïna que augmenta la pressió arterial.
Un altre exemple és la penicil·lina, que inhibeix una proteïna bacteriana essencial per a la síntesi de la paret cel·lular, provocant la mort del bacteri.